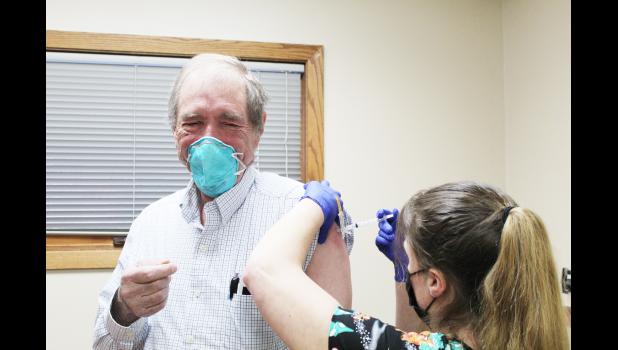
Philip Health Services providers receive COVID-19 vaccine
On Wednesday, December 23, 2020, Philip Health Services (PHS) issued the first two COVID-19 vaccinations to PHS healthcare providers. The vaccine that is being utilized by PHS is the Moderna COVID-19 vaccine, that received emergency use authorization (EUA) from the United States Food and Drug Administration (FDA) on December 18, 2020. The Moderna COVID-19 vaccine is the second vaccine to obtain EUA from the FDA, the first being a COVID-19 vaccine developed by Pfizer/BioNTech. According to FDA Commissioner, Stephen M. Hahn, M.D., “Through the FDA’s open and transparent scientific review process, two COVID-19 vaccines have been authorized in an expedited timeframe while adhering to the rigorous standards for safety, effectiveness, and manufacturing quality needed to support emergency use authorization that the American people have come to expect from the FDA.”
According to the Centers for Disease Control and Prevention’s (CDC) website, the Moderna COVID-19 vaccine has not undergone the same type of review as an FDA- approved or cleared product. FDA may issue an EUA when certain criteria are met, which includes that there are no adequate, approved, and available alternatives. In addition, the FDA decision is based on the totality of the scientific evidence available showing that the product may be effective to prevent COVID-19 during the COVID-19 pandemic and that the known and potential benefits of the product outweigh the known and potential risks of the product. All of these criteria must be met to allow for the product to be used during the COVID-19 pandemic.
PHS Clinic Director, Tara Mitchell, says that 60 doses of the vaccine were administered on Wednesday, December 23 and that another 30 are to be administered this week. This begins phase 1 of the COVID vaccination plan, in accordance with the South Dakota Department of Health’s (SDDOH) and Monument Health’s priority schedule and vaccine allocation. Director Mitchell added that healthcare staff seems optimistic about the vaccine. “I think the healthcare staff were relieved to see there is a light at the end of this dark tunnel and they finally saw a glimpse of hope when employees started to become vaccinated last week,” she said.
The Moderna COVID-19 vaccine comes with two doses, to be taken 28 days apart from one another. Director Mitchell stated that it is unknown at this time when the public may elect to get the vaccine, but she provided a graphic from the SDDOH website (included in this article, titled COVID-19 Vaccine Priority Groups).
The PHS healthcare providers to first receive the vaccine were Dr. Coenraad Klopper and Dr. Terry Henrie, PA-C. Henrie stated that regarding the safety profile, he feels confident in taking the vaccine. “I feel the benefit outweighs any risk,” he said. “As it is an mRNA vaccine and not a live virus vaccine, it has a much greater safety profile,” he added. Henrie went on to say, “I’ve had the unfortunate experience of witnessing many members of our community become very ill. I feel this vaccine allows me to better protect myself, my family and those I'm around at the clinic and hospital.”
According to an article published on December 16, 2020 by Daniella Medina, for the Nashville Tennessean, titled “As COVID-19 vaccine distribution…shots”, there are key facts to understand about both COVID-19 vaccines. These include:
• Neither of the vaccines use the live virus that causes COVID-19
• After being vaccinated, because vaccines do not guarantee a 100% effectiveness, there is a chance you could encounter the virus and get sick, according to the CDC.
• It takes a few weeks for the body to build immunity after vaccination, meaning it’s possible to become infected just before or just after getting vaccinated, according to the CDC>
• According to the CDC, it is not clear whether someone could catch the virus that causes COVID-19 after receiving the vaccination but before immunity is built up, and then pass it on to someone else, whether or not they are asymptomatic.
• According to the CDC, people who have already been infected with COVID-19 “may be advised” to get the vaccine, due to severe health risks associated with the virus and the possibility of reinfection.
• It is unclear how long vaccine-induced immunity will last.
Moderna COVID-19
Vaccine Information
According to the CDC’s website, the Moderna COVID-19 vaccine, named mRNA-1273 and manufactured by ModernaTX, Inc., is a two shot vaccine taken 28 days apart. The vaccine is given in the muscle of the upper arm and does not contain eggs, preservatives or latex. Those who should not get the Moderna COVID-19 vaccine include those who have experienced a severe allergic reaction after a previous dose of this vaccine and those who have experienced a severe allergic reaction to any ingredient of this vaccine. For more information about the Moderna COVID-19 vaccine, please visit https://www.fda.gov/media/144638/download to read about information such as the list of ingredients present in the vaccine, the risks and benefits of receiving the vaccine, pre-vaccination information you should tell your healthcare provider and information regarding side effects of the vaccine.